Advanced Diagnostics for Early Breast Cancer Immune Profiling
Breast Immunocarcinogenic Assay
Clinical Interpretation Service of CARMD-Dx


Breast cancer remains one of the most common cancers worldwide, but its progression is shaped not only by genetic mutations but also by the immune system’s interaction with developing tumors. Subtle molecular and immune changes often appear long before tumors become visible on imaging, providing an opportunity for earlier detection and intervention.
The Breast Immunocarcinogenic Assay analyzes biomarker patterns that reveal immune system activation, tumor-promoting inflammation, and prometastatic signaling. This assay provides clinicians with actionable molecular insights to guide risk assessment, early diagnosis, and personalized care strategies.
Why This Test Matters
Key Benefits
Standard scans can miss early immune and molecular changes. The Breast Immunocarcinogenic Assay detects these disruptions at the molecular level so patients and clinicians can act early.
Early Detection Potential
Identifies immune and molecular signals before traditional imaging or pathology.
Personalized Care
Supports precision medicine by highlighting immune pathways relevant to targeted therapies.
Treatment Monitoring
Tracks biomarker changes during therapy and in remission.
Complementary Diagnostic Value
Enhances mammography and biopsy findings with immune-molecular context.

Physician-Ready Report Included
Results in
10-14 days

Reliable Results You Can Trust
Biomarkers Assessed
The Breast Immunocarcinogenic Assay panel measures a targeted range of biomarkers to detect immune activity, tumor-promoting inflammation, and prometastatic signaling:
Download a Sample Report

How It Works
Process
From registration to results, the process is simple, secure, and stress-free. One blood draw gives you answers that imaging can’t.
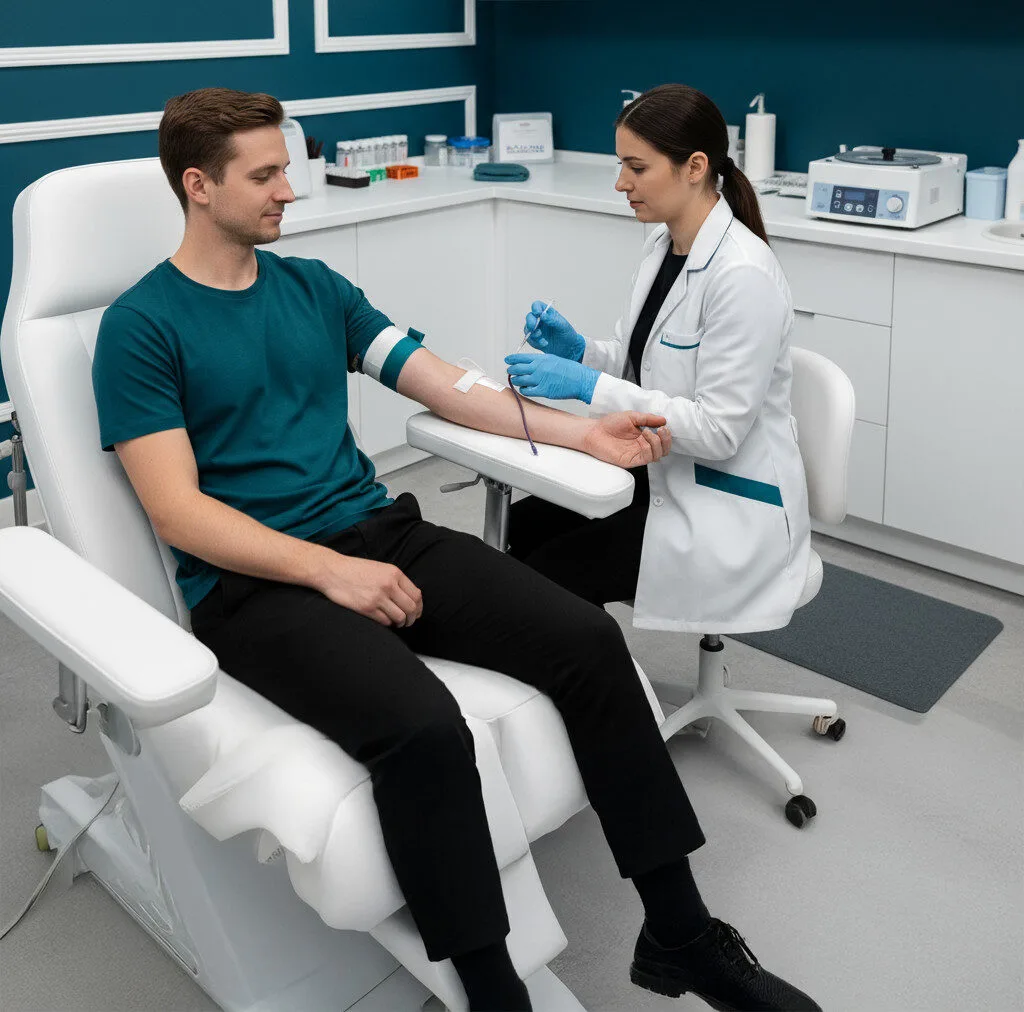
01
Order & Registration
Patient registers through Persona Biomed’s secure portal.
02
Sample Collection
Standard blood draw performed at an approved lab or mobile phlebotomy.
03
Laboratory Analysis
Advanced assays quantify immune and prometastatic biomarker activity.
04
Results Delivery
Detailed physician-ready report delivered via the portal with AI-powered patient summaries.

Who Should Consider This Test
who is this for
This test is intended for individuals who may be at risk, are experiencing related symptoms, or wish to take a proactive approach to their health, such as:

Register Now for
Breast Immunocarcinogenic Assay
Register now for the Breast Immunocarcinogenic Assay to uncover hidden immune signals of breast cancer and take a proactive step toward personalized cancer care
