Advanced Diagnostics for Early Liver Cancer Immune Profiling
Hepatic Prometastatic Reaction Assay
Clinical Interpretation Service of Cancer Testing Services


The liver is a frequent site of both primary cancer development and metastasis from other tumors. Prometastatic molecular activity in the liver microenvironment can create conditions that allow cancerous cells to thrive and spread. Detecting these changes early is critical for preventing progression and improving treatment outcomes.
The Hepatic Prometastatic Reaction Assay analyzes immune-molecular and prometastatic biomarker activity in the liver environment. This test provides early insight into tumor-promoting conditions, helping clinicians anticipate risk, tailor interventions, and monitor long-term liver-related cancer dynamics.
Why This Test Matters
Key Benefits
Standard scans can miss early immune and molecular changes. The Hepatic Prometastatic Reaction Assay detects these disruptions at the molecular level so patients and clinicians can act earl
Early Risk Detection
Identifies immune and prometastatic molecular activity in the liver before overt cancer develops.
Precision Oncology Support
Provides evidence for selecting targeted therapy, immunotherapy, or liver-directed treatments.
Monitoring Tool
Tracks progression, recurrence, or treatment effectiveness in patients with hepatic involvement.
Complementary Evidence
Enhances imaging and biopsy findings with immune-molecular context.

Physician-Ready Report Included
Results in
10-14 days

Reliable Results You Can Trust
Biomarkers Assessed
The Hepatic Prometastatic Reaction Assay panel measures a targeted range of biomarkers to detect immune activity, tumor-promoting inflammation, and liver tumor progression:
Download a Sample Report

How It Works
Process
From registration to results, the process is simple, secure, and stress-free. One blood draw gives you answers that imaging can’t.
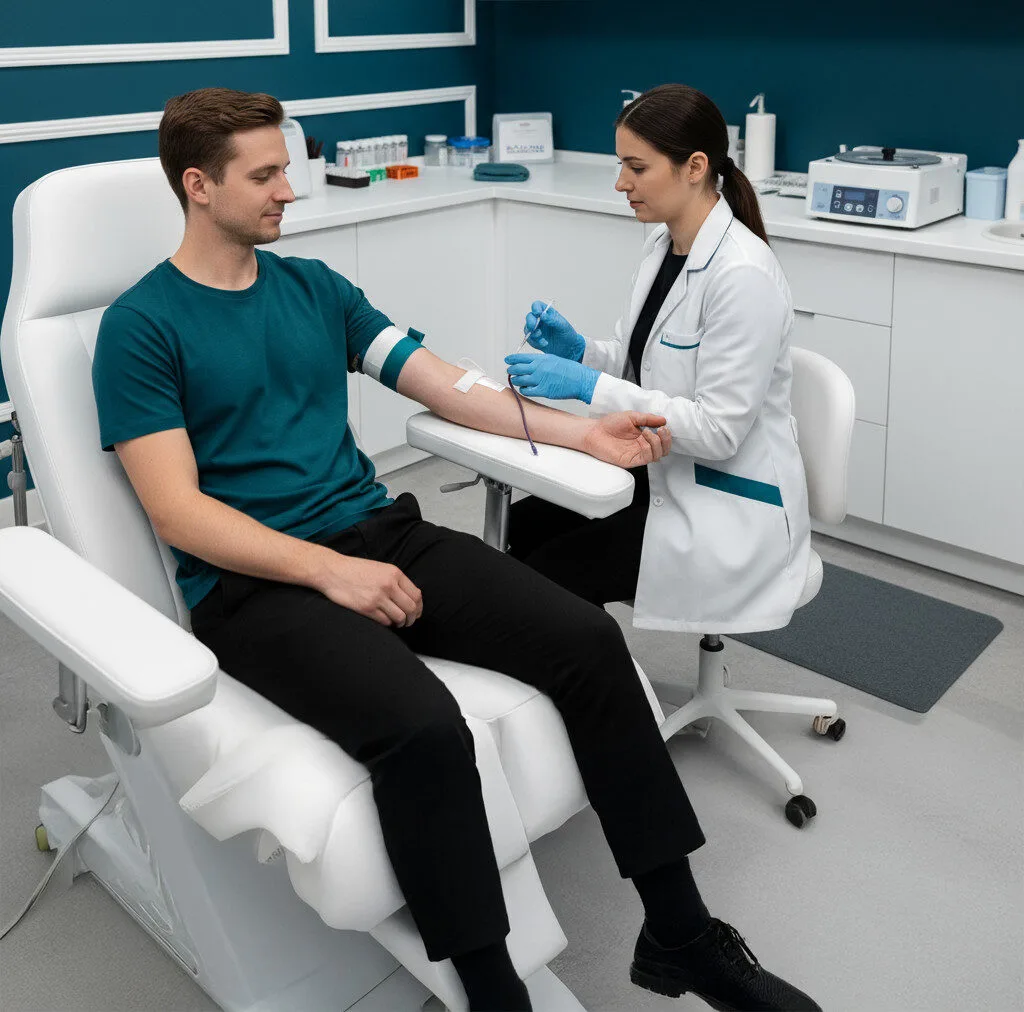
01
Order & Registration
Register securely through Persona Biomed’s patient portal.
02
Sample Collection
Blood draw at a certified site or via mobile phlebotomy.
03
Laboratory Analysis
Biomarker assays evaluate prometastatic immune and metabolic activity in the liver.
04
Results Delivery
Clinical interpretation report delivered through the portal with physician-ready detail and AI-supported summaries.

Who Should Consider This Test
who is this for
This test is intended for individuals who may be at risk, are experiencing related symptoms, or wish to take a proactive approach to their health, such as:

Register Now for Hepatic Prometastatic Reaction Assay
Register now for the Hepatic Prometastatic Reaction Assay to uncover molecular signals of liver-related cancer risk and guide precision care.
